All Five Region Species: 2p3d
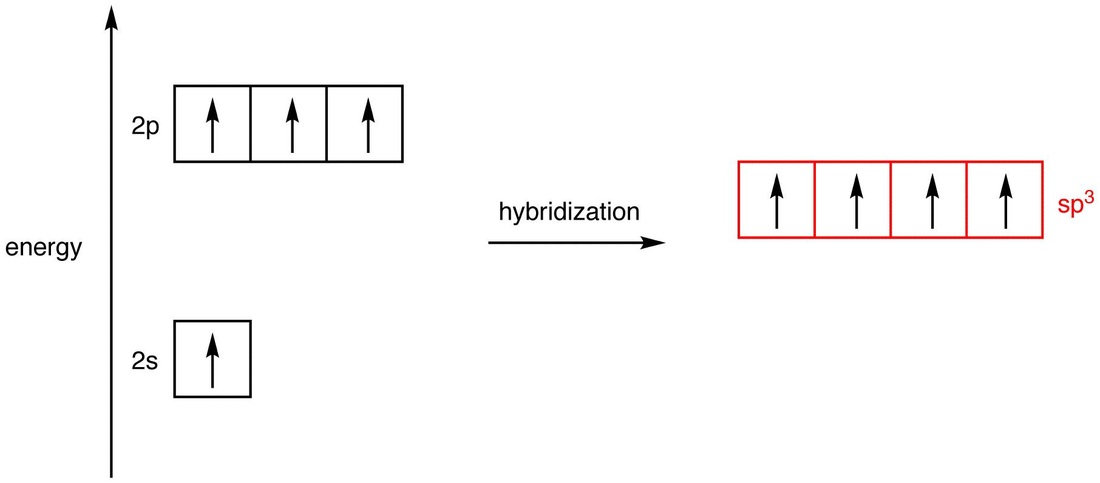
The four hybridized orbitals are created by mixing one 2s orbital and three 2p orbitals. They are called the sp3-hybridized orbitals, and the energy of a sp3-hybridized orbital, on the graph, falls between those of a 2s orbital and a 2p orbital, closer to that of a 2p orbital. The four atomic orbitals used in hybridization had a total of four electrons, which are to be distributed in the four sp3-hybridized orbitals. Since the four sp3-hybridized orbitals have the same energy, according to Hund’s rule, the electrons must be equally distributed in them.
In each sp3-hybridized orbital there’s an electron that repel each other. To minimize the repulsion between electrons, the four sp3-hybridized orbitals arrange themselves around the carbon nucleus so that they are as far away as possible from each other, resulting in the tetrahedral arrangement of the four sp3-hybridized orbitals around the carbon nucleus (5). The carbon atom 5 is called a “sp3-hybridized carbon atom.”
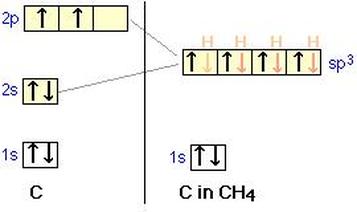
An example of sp3d in Methane (CH4)
-Kyra Mica Ella Flores
-Kyra Mica Ella Flores