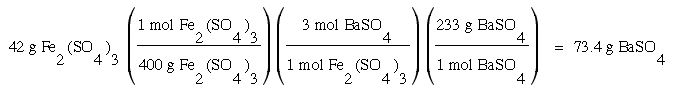Mass to Mass Stoichiometry
Mole to mole stoichiometry is an essential factor in chemistry because it will allow us to convert one substance to another within the limitations of a chemical reaction. So for further knowledge about this stoichiometry, let us imagine this reaction:
1. How many grams of BaSO4 can be made when 42 g of Fe2(SO4)3 is used in the following balanced chemical equation:
Fe2(SO4)3 + 3 BaS --- 3 BaSO4 + Fe2S3
How are you going to solve for this?
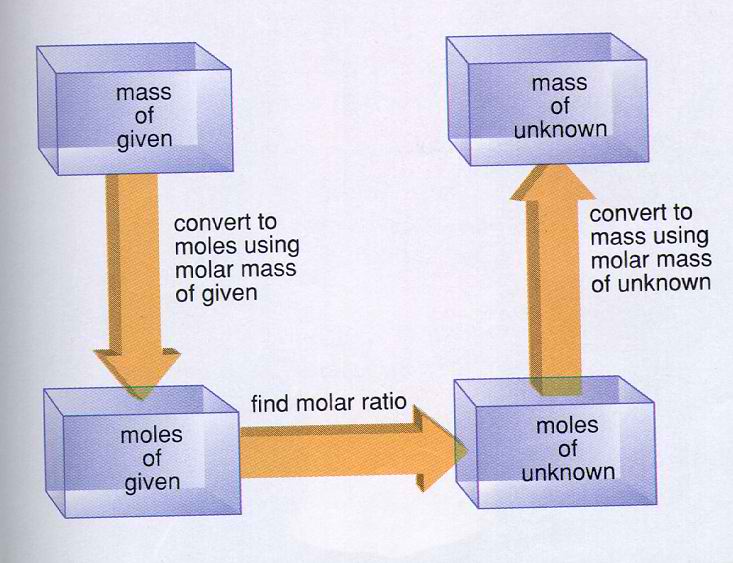
You have some steps to follow to accomplish the mass to mass stoichiometry. See the diagram below:
The answer to the equation above is:
- Liliane Suarez