All Two Region Species: 2sp
Consider the Lewis structure of gaseous molecules of BeF2:
We can see that in Fluorine’s orbital structure there is an unpaired electron in the 2p orbital. So, it means that the unpaired electron in the 2p orbital can be paired in an electron to form a covalent bond.
Let us work now in the Beryllium’s orbital structure.
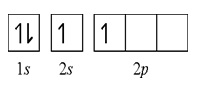
I’m sure that you can clearly see that there are no unpaired electrons in the orbital structure of the Beryllium atom. That means that the Beryllium atom is incapable of forming a covalent bond with a fluorine atom. However, the Beryllium atom can obtain an unpaired electron by promoting an electron from the 2s orbital to the 2p orbital. Remember that when we say promotion we are after in putting the said electron to a higher energy level. So from 2s we can promote the other electron to 2p because 2p is greater in energy than the 2s. Now the structure would be like this:
This would actually result in two unpaired electrons, one in a 2s orbital and another in a 2p orbital.
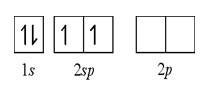
The two unpaired orbital (2s and 2p) will be combined to form a newly hybrid orbital. And we will call it as 2sp hybridization. And the final structure would be this:
The two unpaired orbital (2s and 2p) will be combined to form a newly hybrid orbital. And we will call it as 2sp hybridization. And the final structure would be this:
The Beryllium atom is now capable of forming a covalent bond with fluorine. A sigma bond will be formed.
Note that 2sp hybridization can be formed only in a linear geometrical figure.
- Jen Odessa Constantino
Note that 2sp hybridization can be formed only in a linear geometrical figure.
- Jen Odessa Constantino