All Six Region Species: sp3d2
Hybridization of pure atomic orbitals is observed before the
bond formation to confer maximum stability to the molecule. One type of
hybridization is the sp3d2 hybridization.
For example:
Sulfur hexafluoride (SF6)
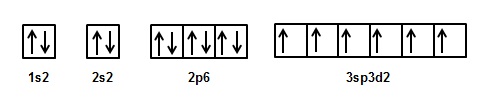
The electronic configuration of 'S' in ground state is 1s2 2s2 2p6 3s2 3px2 3py1 3pz1.
In the SF6 molecule, there are six bonds formed by sulfur atom. Hence there must be 6 unpaired electrons. However there are only 2 unpaired electrons in the ground state of sulfur. Hence it promotes two electrons into two of the 3d orbitals (one from 3s and one from 3px).
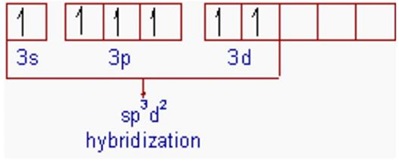
Thus the electronic configuration of 'S' in its 2nd excited state is 1s2 2s2 2p6 3s1 3px1 3py1 3pz1 3d2.
For example:
Sulfur hexafluoride (SF6)
The electronic configuration of 'S' in ground state is 1s2 2s2 2p6 3s2 3px2 3py1 3pz1.
In the SF6 molecule, there are six bonds formed by sulfur atom. Hence there must be 6 unpaired electrons. However there are only 2 unpaired electrons in the ground state of sulfur. Hence it promotes two electrons into two of the 3d orbitals (one from 3s and one from 3px).
Thus the electronic configuration of 'S' in its 2nd excited state is 1s2 2s2 2p6 3s1 3px1 3py1 3pz1 3d2.
In the second excited state, sulfur under goes sp3d2 hybridization by mixing a 3s, three 3p and two 3d orbitals. Thus formed six half-filled sp3d2 hybrid orbitals are arranged in octahedral symmetry.
Sulfur atom forms six sp3d2-p bonds with 6 fluorine atoms by using these sp3d2 orbitals. Each fluorine atom uses is half-filled 2pz orbitals for the bond formation.
Sulfur atom forms six sp3d2-p bonds with 6 fluorine atoms by using these sp3d2 orbitals. Each fluorine atom uses is half-filled 2pz orbitals for the bond formation.
- Leonardo Gabriel Lira