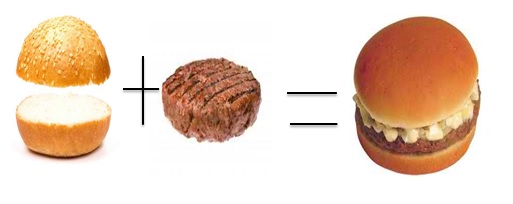Limiting and Excess Reactants
Let us understand the differences of excess reactant and limiting reactant in a chemical reaction. In this article you can gain knowledge about limiting and excess reactants and it will help you to undertsand their differences.
Suppose that you are a cook in a Burger restaurant .In order to make a regular burger, you need 2 buns and 1 burger patty (just neglect the other ingredients). In this problem, visualize that the buns and burger patty are the reactants while the burger is the product form the reaction of 2 buns and 1 burger patty.If you have 30 pieces of buns and 12 pieces of burger patty, how many burger can you make? Using 30 pieces of buns, you can produce 15 burgers and using 12 patties you can produce 12 burgers. Although you can make more burgers from the amount of burger buns that are available,you can only create 12 burgers because of thye number of burger patties available, making the burger pattiesthe limiting reagent in this reaction. The buns are the excess because the number of burgers formed by 30 buns produced by 12 patties, the patties are the limiting reactant.
Suppose that you are a cook in a Burger restaurant .In order to make a regular burger, you need 2 buns and 1 burger patty (just neglect the other ingredients). In this problem, visualize that the buns and burger patty are the reactants while the burger is the product form the reaction of 2 buns and 1 burger patty.If you have 30 pieces of buns and 12 pieces of burger patty, how many burger can you make? Using 30 pieces of buns, you can produce 15 burgers and using 12 patties you can produce 12 burgers. Although you can make more burgers from the amount of burger buns that are available,you can only create 12 burgers because of thye number of burger patties available, making the burger pattiesthe limiting reagent in this reaction. The buns are the excess because the number of burgers formed by 30 buns produced by 12 patties, the patties are the limiting reactant.
Therefore:
The LIMITING REACTANT is the substance that limits the amount of product that will be produce and the reactant that gives the maximum amount of product in the reaction.
The EXCESS REACTANT is the substance that is excess because of the limiting reactant and the reactant/reagent that gives the excess amount in a reaction.
The COMPLETION is when there will be product formation. To solve the problem you must identify which of the reactant is going to run out.
To Find the Limiting Reagent
There are two main ways to determine the limiting reagent. One way is to find and compare the mole ratio of the amount of reactants used in the reaction (see formula 1). Another way is to calculate the grams of products produced from the given quantities of reactants; the reactant that produces the least amount of product is the limiting reagent (see formula 2).
Formula 1: Find the limiting reagent by looking at the number of moles of each reactant.
- Determine the balanced chemical equation for the chemical reaction.
- Convert all given information into moles (most likely, through the use of molar mass as a conversion factor).
- Calculate the mole ratio from the given information. Compare the calculated ratio to the actual ratio.
- Use the amount of limiting reactant to calculate the amount of product produced.
- If necessary, calculate how much is left in excess of the non-limiting reagent.
Formula 2: Find the limiting reagent by calculating and comparing the amount of product each reactant will produce.
- Balance the chemical equation for the chemical reaction.
- Convert the given information into moles.
- Use stoichiometry for each individual reactant to find the mass of product produced.
- The reactant that produces a lesser amount of product is the limiting reagent.
- The reactant that produces a higher amount of product is the excess reagent.
- To find the amount of remaining excess reactant, subtract the mass of excess reagent consumed from the total mass of excess reagent given.
Examples:
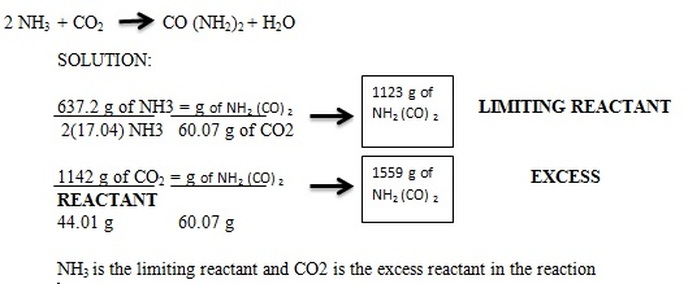
What would be the limiting reagent if 78 grams of Na2O2 were reacted with 29.4 grams of H2O?
Using Formula 1:
A. 78g x (1 mol/77.96g) = 1.001 moles of Na2O2
29.4g x (1 mol/18g)= 1.633 moles of H2O
B. If we assume that all of the water is used up, 1.633 x (2/2) or 1.633 moles of Na2O2 are required. Since there are only 1.001 moles of Na2O2, it is the limiting reactant.
Using Formula 2:
78g Na2O2 x (1 mol Na2O2)/(77.96g Na2O2) x (4 mol NaOH)/(2 mol Na2O2) x (40g NaOH)/(1 mol NaOH) = 80.04g NaOH
Therefore, we find that using either formulas yield Na2O2 as the limiting reagent.
Which of the following reactant is the limiting in the reaction?
- Charisse Brittney Ann Vicente