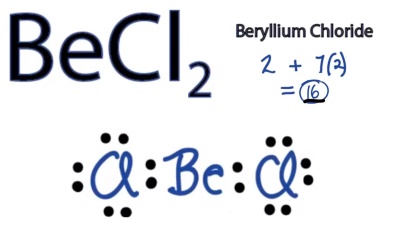Too Few Electron
Hydrogen, beryllium and boron have too few electrons to form an octet. Hydrogen has only one valence electron, and only one place to form a bond with another atom. Beryllium only has two valence atoms, and can only form electron pair bonds in two locations. Boron has three valence electrons. The two molecules depicted in this picture show the central beryllium and boron atoms with fewer than eight valence electrons.
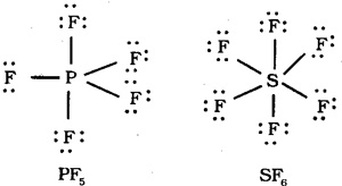
Too Many Electron
Elements in periods greater than
period 3 on the periodic table have a d
orbital available with the same
energy quantum number. Atoms in these periods may follow the octet rule, but
there are conditions where they can expand their valence shells to accommodate
more than eight electrons.
Lonely Electrons
Most stable molecules and complex
ions contain pairs of electrons. There is a class of compounds where the
valence electrons contain an odd number of electrons in the valence shell.
These molecules are known as free radicals. Free radicals contain at least one
unpaired electron in their valence shell. In general, molecules with an odd
number of electrons tend to be free radicals.
- Laurenz Xilec Lim