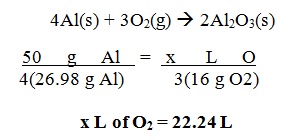Mass to Volume Stoichiometry
In the case of mass-volume stoichiometry, either the mass of a compound will be given and the volume of another is asked, or the volume of a gas will be given and the mass of another compound will be asked.
It's important to understand the concept of STP (standard temperature and pressure) in order to answer mass-volume stoichiometry problems. Standard temperature and pressure is a set of conditions wherein the temperature is equal to 273.15 K and the pressure is equal to 1 atm at which 1 mole of any ideal gas will occupy a volume of 22.414 L.
For example:
Calculate the volume (in liters) of oxygen gas required to react with 50 g of aluminum at STP.
Ø You must first write the CORRECT chemical equation then balance it.
Ø Solve for the formula mass of the given elements / compounds.
Ø Then, perform the cross multiplication method to get the answer.
- Kyra Mica Ella Flores