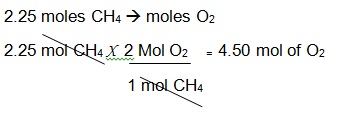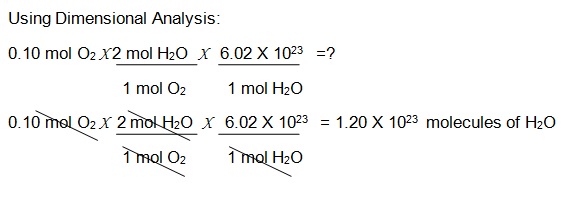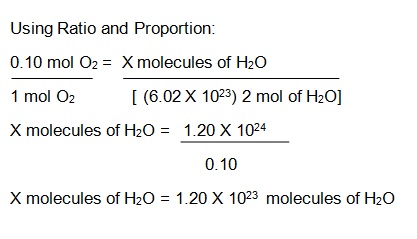Mole to Mole Stoichiometry
When we say mole to mole stoichiometry we are referring to a type of calculation that relates the mole of two substances participating in a balanced chemical equation.
The coefficients in a balanced chemical equation can be used to determine the relative number of molecules, formula units, or moles of a compound involved in a chemical reaction.
In performing mole to mole stoichiometry the first thing we need to do is to balance a chemical reaction.
Example, we are given a chemical reaction CH4 + O2 à CO2 + H20
Note that the given chemical reaction is unbalanced and the balanced equation is CH4 + 2 O2 à CO2 + H20 (1 molecule of Methane is needed to react with 2 moles of Oxygen to produce 1 mole of Carbon dioxide and 1 mole of Water) We can now act upon the mole to mole stoichiometry.
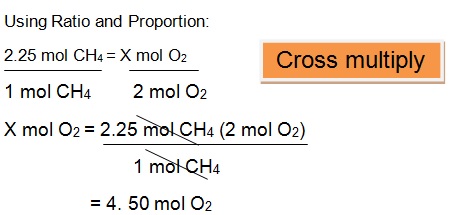
Let us say we are looking for the moles of oxygen reacting with 2.25 moles of Methane.
Proportion of Methane to Oxygen: 1:2
Using Dimensional Analysis:
Since we have 2.25 mol of CH4 , and a proportion of 1:2. Divide the 2.25 mol of CH4 by 1 mol of CH4 to cancel the unit mol CH4 and multiply it by 2 moles of O2 to come up with an answer of 4.50 moles of O2.
If you were ask to find molecules of Water in the reaction
02 + H2 = H20, and you have 0.10 mol of 02 just use the Avogadro’s number. Remember that the Avogadro constant is defined as the number of constituent particles, atom, or formula unit per mole of a given substance.
Balanced Equation: 02 +2 H2 = 2 H20 (1 mol of oxygen is needed to react with 2 moles of Hydrogen to form 2 moles of Water)
Proportion of Oxygen to Water: 1:2
02 + H2 = H20, and you have 0.10 mol of 02 just use the Avogadro’s number. Remember that the Avogadro constant is defined as the number of constituent particles, atom, or formula unit per mole of a given substance.
Balanced Equation: 02 +2 H2 = 2 H20 (1 mol of oxygen is needed to react with 2 moles of Hydrogen to form 2 moles of Water)
Proportion of Oxygen to Water: 1:2