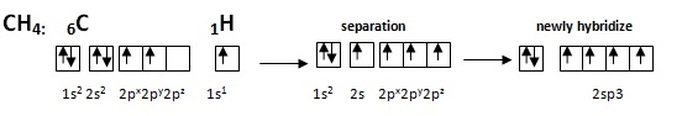
hybridization: four region species (sp3)
Overlap must occur between the orbitals containing the valence electrons in order for a covalent bond to form between 2 atoms.
We are now having the four region species or the 2sp3. In order for the best overlap to occur, valence electrons need to be re-oriented. CH4 is a good example for this.
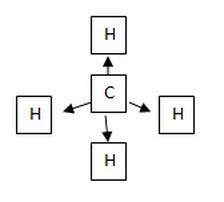
This will result into a tetrahedral structure:
-Krysta Coleen Matias